
Indian Prime Minister Narendra Modi on Thursday said New Delhi will deny Pakistan access to water resources from Indian territory because of Islamabad’s hostile policies.
“Pakistan will not get a drop of water that belongs to India. Playing games with Indian lives will cost Pakistan dearly,” Hindustan Times quoted the prime minister as saying.
He also recalled New Delhi’s stance that talks with Islamabad would be held only on the Pakistan-controlled part of Kashmir.
Earlier it was reported that the Indian side, despite the truce with Islamabad after the exchange of blows in May, has not yet returned to the implementation of the Indus Waters Treaty, which regulates the use of water from several rivers by the parties.
Meanwhile, according to Dawn, Bilawal Bhutto Zardari, chairman of one of the country’s ruling Pakistan People’s Party, said the previous day that peace in South Asia could be achieved if only India and Pakistan agreed on three issues – Kashmir, counter-terrorism and water.
“The people of the two countries desire peace which is not possible without solving the three issues of Kashmir, terrorism and water,” he said.
The politician also expressed deep concern over India’s exploitation of the water issue for its own interests.
Dawn notes that Zardari, at the head of a Pakistani delegation, will soon travel to a number of countries to convey Islamabad’s views on the regional situation.
The treaty, concluded in 1960, governs the use by both countries of water from the Indus and several other rivers that originate in India. In a new round of tensions between the countries that began in April 2025, India suspended implementation of the treaty and closed the locks on some river dams. Islamabad fears that such actions could cause drought in Pakistani territory.

Against the backdrop of escalating tensions in Kashmir in May 2025, India and Pakistan once again found themselves on the brink of military conflict. After a terrorist attack in Pahalgam that killed 26 people, India accused Pakistani militants of being responsible, which led to a sharp deterioration in relations between the two countries. Pakistan, in turn, rejected the accusations and conducted missile tests, demonstrating its readiness to take appropriate measures.
Let’s take a look at the military capabilities of these two countries.
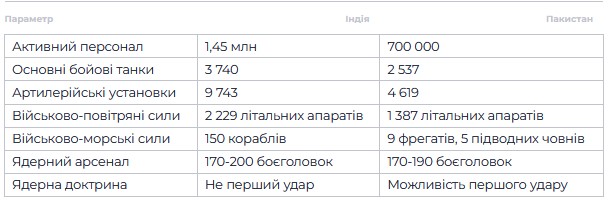
India’s military capabilities
Troop strength: 1.45 million active military personnel, 1.15 million reservists, and over 2.5 million paramilitary personnel.
Ground forces: 3,740 main battle tanks (T-90 Bhishma, Arjun), 9,743 artillery units, including modern Pinaka and Dhanush systems.
Air force: approximately 2,229 aircraft, including 600 fighter jets (Rafale, Su-30MKI), 899 helicopters, and 831 support aircraft.
Navy: 75,500 personnel, 150 ships, including the aircraft carrier INS Vikramaditya and Arihant-class ballistic missile submarines.
Nuclear arsenal: approximately 170–200 warheads, a “no first strike” policy, delivery systems — Agni (I–V), Prithvi, BrahMos missiles, as well as sea and air platforms.
Technological achievements: development of hypersonic missiles (BM-04), laser air defense systems (IDD&IS Mk2A), integration of theater commands, and the Integrated Battle Groups concept for rapid mobilization.
Pakistan’s military potential
Number: approximately 700,000 active military personnel: 560,000 in the army, 70,000 in the air force, and 30,000 in the navy.
Army: 2,537 main battle tanks (T-80, Al-Khalid), 4,619 artillery pieces.
Air Force: approximately 1,387 aircraft, including JF-17 Thunder and F-16 fighter jets, as well as Chinese drones.
Navy: 30,000 personnel, 9 frigates, 5 submarines, including new Chinese S20s.
Nuclear arsenal: approximately 170–190 warheads, a “first strike” doctrine with an emphasis on tactical nuclear weapons, including Nasr (Hatf-9) missiles with a range of up to 70 km.
Technological achievements: joint programs with China for the production of fighter jets, modernization of air defense, development of missile technologies.
The situation remains tense:
Pakistan has conducted two missile tests, including the launch of a Fatah missile with a range of 120 km.
India has stepped up security measures and conducted exercises in several states.
Both countries have closed their airspace and suspended trade.
International actors are calling for de-escalation.
Experts warn that even a limited conflict could escalate into a nuclear confrontation, given the doctrines of the parties and historical experience.

Indian military officials said Pakistani troops fired on Indian posts along the Line of Control in disputed Kashmir for the second time in a night, the Associated Press reported on Saturday.
According to a statement from the Indian Army, soldiers from several Pakistani army posts opened fire on Indian positions “across the Line of Control.”
Indian troops responded with small arms fire. The statement said the firing was ‘unprovoked,’ but no information on casualties was provided.
Tensions between India and Pakistan have escalated after a deadly attack on Indian tourists in Kashmir on Tuesday, when a terrorist attack killed about 30 people, most of whom were Indian citizens.
India called the attack a “terrorist attack” and accused Pakistan of supporting the attackers. The Indian Express, citing sources, reported that seven attackers were behind the attack, at least four of whom were from Pakistan.
Pakistan, in turn, denied any involvement, and an unknown militant group called the “Kashmir Resistance” claimed responsibility for the attack.
In response to the escalation of the conflict, India suspended an important agreement on the joint use of water resources and closed the only functioning land border crossing point.
Pakistan, in turn, canceled visas for Indian citizens and closed its airspace to Indian airlines.

The Interdepartmental Commission on International Trade (ICIT) imposed a final anti-dumping duty of 35.7% on imports of household matches to Ukraine on April 16.
According to the ICIT’s announcement in the Uryadovyi Kurier newspaper on Friday, the decision will enter into force 30 days after the date of publication of the announcement.
The ICIT reminds that the anti-dumping investigation was initiated by the decision of April 12 against imports of matches from India and Pakistan at the complaint of Ukrainian Match Factory LLC, which has a share of more than 50% in the total production of matches in Ukraine.
At the same time, it was found that during the investigation period (April 1, 2023 – March 31, 2024), imports of matches from India were carried out at dumping prices, while there were no imports from Pakistan.
The ICIT found that during the study period (January 1, 2021 – March 31, 2024), the volume of dumped imports decreased by 47.3% in absolute terms, while increasing by 27.8% in relation to the total production of the product in Ukraine and by 26.4% in relation to consumption.
In addition, the national producer’s production volumes decreased by 58.7%, the level of production capacity utilization by 58.7%, domestic sales by 58.1%, financial result from domestic sales in dollar terms by 229.4%, in hryvnia terms by 275.3%, and profitability from domestic sales by 390.4%, which was negative.
The number of production workers also decreased by 2.4%, wages by 32.1%, labor productivity by 57.7%, investment by 100%, and warehouse balances increased by 134.1%.
The Commission found that the Republic of India has a significant export potential, which indicates the likelihood of a significant increase in the volume of dumped imports of matches from this country in the future.
The anti-dumping measures are applied to matches, except for pyrotechnic products of heading 3604, classified under code 3605 00 00 00 according to the Ukrainian Classification of Goods for Foreign Economic Activity.
Ukrainian Match Factory LLC was founded in 1995, with production facilities located in Rivne region. The design capacity of the factory is 630 million boxes of matches per year. The products are supplied to all regions of Ukraine and abroad.
According to opendatabot, in 2024, the factory suffered a loss of almost UAH 26 million (3% more than a year earlier), while net income increased by 9.7% to UAH 175.2 million. At the same time, in 2022, it had a profit of UAH 38 million and revenue of UAH 373.5 million.
The founder of the company is listed as British Paxstone Limited, and the ultimate beneficiary is Maria Fursina from Kyiv.

India has opened its market to apples from Ukraine, the head of the State Service of Ukraine for Food Safety and Consumer Protection, Serhiy Tkachuk, said on Facebook.
“We have important news in the context of opening new foreign markets for the export of Ukrainian agricultural products. Recently, the first trial batch of Ukrainian apples arrived in the Republic of India. The Ministry of
Agriculture and Farmers’ Welfare of India has confirmed the successful completion of the inspection activities of this batch. The Indian side highly appreciated the quality of Ukrainian products and their compliance with the established requirements,” he wrote on Monday.
The head of the State Service of Ukraine for Food Safety and Consumer Protection noted that foreign partners positively assessed compliance with the criteria and requirements for the selection, pre-shipment storage, processing and transportation of Ukrainian agricultural products to India.
Tkachuk thanked the Indian side for the fruitful cooperation and expressed hope that it would continue.
The Head of the State Service of Ukraine for Food Safety and Consumer Protection assured that the agency, together with the Ministry of Foreign Affairs, will continue to work to expand opportunities for Ukrainian producers. “Support for the state’s economy and Ukrainian exporters is vital in the context of a full-scale war,” he summarized.

Rice prices fell after India lifted the last of its existing restrictions on rice exports.
The price per ton of benchmark white Thai rice, which was $669 in January 2024, had fallen to $405 by last week, the Financial Times said. The decision to lift the restrictions was prompted by India’s desire to boost agricultural and food exports to boost farmers’ incomes amid a general economic slowdown in the country.
According to Commerce Minister Piyush Goyal, the plan is to increase shipments to $100 billion by 2030 from $48.15 billion in 2023-2024.
“Last year, about $50 billion worth of products were exported from India,” the minister had said earlier. – I hope to see a triple-digit figure, the $100 billion mark.”
Export restrictions were imposed in the country in 2022. As a result of the decision, the price of white Thai rice jumped to its highest since 2008. India began easing the restrictive measures in September last year. India’s rice exports, which stood at 14 million tons in 2023, could reach a record 21.5 million tons between September 2024 and October 2025, according to S&P Global estimates.
The return of Indian rice will negatively impact exporters from Pakistan, which has gained market shares in Indonesia and East Africa amid declining supplies from India. The US Department of Agriculture estimates rice exports from Pakistan for the 12 months to May 2025 at just 5.8 million tons, down 11.4% from the same period a year earlier.
India is a leading supplier of milled rice, which is in high demand in African countries. According to the International Food Policy Research Institute, Indian supplies accounted for more than 60% of rice imports of 17 African countries and more than 80% of imports of nine, including Somalia, in 2022.